これって本当ですか?
「ロタワクチンがロタウイルスを排除したので、ノロウイルスが脅威を奮うようになった」
もうひとつあります
「水疱瘡のワクチンで水疱瘡の発症が減少したので、大人の帯状疱疹が増えた
帯状疱疹は小児でも発症するようになり、ベテランの小児科医を驚かせている」
これって本当ですか?
「ロタワクチンがロタウイルスを排除したので、ノロウイルスが脅威を奮うようになった」
もうひとつあります
「水疱瘡のワクチンで水疱瘡の発症が減少したので、大人の帯状疱疹が増えた
帯状疱疹は小児でも発症するようになり、ベテランの小児科医を驚かせている」
Published: 6 December, 2013
By ALICE HUTTON
A WORLD-famous cancer scientist from Kilburn whose work was “nothing short of historic” died in unexpected and mysterious circumstances inside a locked room, an inquest has heard.
Dr Anne Szarewski’s pioneering discovery of the cause of cervical cancer led to the first-ever HPV vaccine, saving the lives of countless women across the globe.
記事要約
2013年8月24日、世界的に有名ながん研究者のスザルゼウスキー医師が自宅で死亡しているのを、夫により発見された。
ロンドンの大学に所属、子宮頸がんワクチンの開発に多大な貢献をした。ガン研究所のコンサルタントもしていた。
死因は、判定不能であったが、血液からクロロキン(https://ja.wikipedia.org/wiki/%E3%82%AF%E3%83%AD%E3%83%AD%E3%82%AD%E3%83%B3)が検出されている。自宅の薬棚にはこの薬剤があったが、医師は大量の薬剤のコレクションを所有していたので、服薬自殺とは断定されていない。

Cervical cancer is the third most common cancer in women worldwide and often affects women under 40 years of age with young families. Vaccination against HPV is a major advancement, as it offers primary prevention against the infectious agent that is the main cause of the disease. The bivalent AS04-adjuvanted prophylactic HPV vaccine provides high efficacy against disease associated with HPV 16 and 18, as well as significant cross-protection against some HPV types not included in the vaccine. Protection against HPV 45 may be particularly important, as it is relatively more common in adenocarcinoma. The vaccine's antibody response profile suggests a long duration of immunity. Safety data have been reassuring, which is not unexpected, given that the vaccine is composed of virus-like particles, rather than being a live-virus vaccine.
これって本当なのだろうか
・猫ひっかき病が、乳がんの原因となることがある
https://www.hindawi.com/journals/crionm/2013/507504/
https://infectagentscancer.biomedcentral.com/articles/10.1186/s13027-015-0025-x
ガーダシルの後、バルトネラが陽性だったケース 2例あり
| VAERS ID: | 350414 (history) |
| Form: | Version 1.0 |
| Age: | 19.0 |
| Gender: | Female |
| Location: | California |
| Vaccinated: | 2009-01-20 |
| Onset: | 2009-01-20 |
| Days after vaccination: | 0 |
| Submitted: | 2009-06-29 |
| Days after onset: | 159 |
| Entered: | 2009-06-29 |
| Vaccination / Manufacturer | Lot / Dose | Site / Route |
| HPV4: HPV (GARDASIL) / MERCK & CO. INC. | 1311X / 3 | UN / UN |
| MMR: MEASLES + MUMPS + RUBELLA (MMR II) / MERCK & CO. INC. | 0812U / UNK | UN / UN |
| VARCEL: VARICELLA (VARIVAX) / MERCK & CO. INC. | 0270 / UNK | UN / UN |
Administered by: Private Purchased by: Unknown
Symptoms: Asthenopia, Back pain, Bartonella test positive, Eye inflammation, Eye operation, Eyelid ptosis, Headache, Intraocular pressure increased, Laboratory test, Neck pain, Photophobia, Photopsia, Retinal exudates, Retinal haemorrhage, Retinal vasculitis, Retinitis, Vision blurred
SMQs:, Haemorrhage terms (excl laboratory terms) (narrow), Anticholinergic syndrome (broad), Retroperitoneal fibrosis (broad), Noninfectious meningitis (narrow), Glaucoma (narrow), Optic nerve disorders (broad), Lens disorders (broad), Corneal disorders (broad), Retinal disorders (narrow), Vasculitis (narrow), Periorbital and eyelid disorders (narrow), Ocular infections (narrow), Ocular motility disorders (narrow), Arthritis (broad), Hypoglycaemia (broad)
Life Threatening? No
Birth Defect? No
Died? No
Permanent Disability? Yes
Recovered? No
Office Visit? No
ER Visit? Yes
ER or Doctor Visit? No
Hospitalized? No
Previous Vaccinations:
Other Medications:
Current Illness: None
Preexisting Conditions: 6/30/09 Received medical records PMH: appendectomy, small bowel resection, tonsillectomy. Medical records received 7/7/09 PMH: motor vehicle accident, pleurisy, sulfa allergy
Allergies:
Diagnostic Lab Data: Many-being treated for last 5 months. Medical records received 7/7/09 LABS: Bartonella benselae (+)
CDC Split Type:
Write-up: Pt c/o headache, the began c/o sensitivity to light. On 1/22/09 went to have eyes checked retinas inflamed. Went to ophthalmologist retinas being treated. Then pressure went up, and had surgery on R eye 6/23/09. 7/7/09 Consultant records received DOS 2/4/09 to 6/2/09. Assessment: Retinal vasculitis and vitritis, pars planitis, increased ocular pressure. Patient presents with flashes left eye, black lines. Headaches during day, eyes light sensitive. Floaters right eye. 8/24/09 Received Ophtho medical records of 6/9/09-8/18/09. FINAL DX: Pars planitis right eye; increased intraocular pressure. Patient tx w/diuretic & drops. IOP remained high, developed severe HE.. Surgery of trabeculectomy w/antimetabolites, right eye done 6/23/09. continued to have pain & blurred vision post operatively. Developed heavy feeling in right eye, increased floaters in left eye & hazy vision. 6/30/09 Received PCP & vaccine records of 1/20/2009-2/4/09. Records reveal patient experienced back ache, cervicalgia, HA. Received depo-provera & vaccines. Referred to retina specialist on 2/3 but no office note. Follow-up: Pt has had 2 surgeries on her right eye. She will have 1 more next month. Her right eye droops, blocking her vision, sensitive to light.
| VAERS ID: | 572528 (history) |
| Form: | Version 1.0 |
| Age: | 11.0 |
| Gender: | Female |
| Location: | Unknown |
| Vaccinated: | 2010-09-02 |
| Onset: | 0000-00-00 |
| Submitted: | 2015-04-01 |
| Entered: | 2015-04-01 |
| Vaccination / Manufacturer | Lot / Dose | Site / Route |
| HPV4: HPV (GARDASIL) / MERCK & CO. INC. | - / 1 | UN / UN |
Administered by: Other Purchased by: Other
Symptoms: Abdominal pain, Activities of daily living impaired, Arthralgia, Asthenia, Autoimmune disorder, Autonomic nervous system imbalance, Bartonella test positive, Blood test, Borrelia test negative, Coagulation test abnormal, Cognitive disorder, Computerised tomogram, Dark circles under eyes, Dizziness, Ear pain, Fatigue, Feeling abnormal, Formication, Headache, Human herpesvirus 6 infection, Hyperacusis, Hypersensitivity, Hypersomnia, Hypoaesthesia, Hypocoagulable state, Immune system disorder, Immunology test abnormal, Listless, Lyme disease, Malaise, Memory impairment, Migraine, Mycoplasma test positive, Mycotic allergy, Nausea, Neuralgia, Nuclear magnetic resonance imaging, Oropharyngeal pain, Pain, Pallor, Palpitations, Paraesthesia, Photophobia, Pneumonia mycoplasmal, Psychosomatic disease, Roseolovirus test positive, Skin striae, Streptococcal infection, Streptococcus test positive, Syncope, Thyroid disorder, Vaccination complication, Visual impairment
SMQs:, Torsade de pointes/QT prolongation (broad), Liver-related coagulation and bleeding disturbances (narrow), Acute pancreatitis (broad), Angioedema (broad), Peripheral neuropathy (narrow), Haemorrhage laboratory terms (broad), Neuroleptic malignant syndrome (broad), Anticholinergic syndrome (broad), Arrhythmia related investigations, signs and symptoms (broad), Retroperitoneal fibrosis (broad), Dementia (broad), Oropharyngeal conditions (excl neoplasms, infections and allergies) (narrow), Guillain-Barre syndrome (broad), Noninfectious encephalitis (broad), Noninfectious encephalopathy/delirium (broad), Noninfectious meningitis (narrow), Gastrointestinal nonspecific symptoms and therapeutic procedures (narrow), Glaucoma (broad), Optic nerve disorders (broad), Cardiomyopathy (broad), Lens disorders (broad), Corneal disorders (broad), Retinal disorders (broad), Hypothyroidism (broad), Hyperthyroidism (broad), Depression (excl suicide and self injury) (broad), Hearing impairment (narrow), Vestibular disorders (broad), Hypotonic-hyporesponsive episode (broad), Hypersensitivity (narrow), Arthritis (broad), Drug reaction with eosinophilia and systemic symptoms syndrome (broad), Hypoglycaemia (broad), Infective pneumonia (narrow)
Life Threatening? No
Birth Defect? No
Died? No
Permanent Disability? Yes
Recovered? No
Office Visit? No
ER Visit? Yes
ER or Doctor Visit? No
Hospitalized? No
Previous Vaccinations:
Other Medications: No other medications
Current Illness: Unknown
Preexisting Conditions:
Allergies:
Diagnostic Lab Data: Lyme disease test (OCT-2010): Negative. Bartonella henselae test (MAY-2011): Highly positive. Mycoplasma pneumoniae test (MAY-2011): Highly positive. Lyme disease test (MAY-2011): Negative. Strep test (JUN-2011): Chronic strep (positive). Viral test (JUN-2011): Human herpes virus 6 (HHV6, positive). Coagulation test (JUN-2011): Hypocoagulation (decreased). 10/2010, Borrelia test, Negative; 05/2011, Borrelia test, Negative; 05/2011, Laboratory test, Her immune system was struggling
CDC Split Type: WAES1503USA014913
Write-up: This spontaneous report as received from the patient''s mother in an article online via a company representative, refers to a 11 year old female patient who was normal, conscientious, healthy and very active. The patient''s medical history was not reported. On 02-SEP-2010, the patient was vaccinated with her first dose of GARDASIL (dose, lot number and route not provided). No co-suspect or concomitant therapies were provided. The patient''s mother stated that on 07-SEP-2010 was the patient''s first day of middle school and approximately in September 2010, after receiving GARDASIL, the patient began to experience fatigue and headaches. The patient did not feel good. She began sleeping over 12 hours a day and even more on the weekends. She missed days at school, dance lessons and cheer practices. Soon, her illness was visible on the outside too. She did not look good, she had circles under her eyes, her skin color was ashen and she appeared listless. Approximately in September 2010, the patient''s earliest symptoms were a constant headache or migraine that did not respond to pain relievers, stabbing 24/7 bilateral ear pain, fatigue not relieved by sleep, abdominal pain, nausea and joint pain. The patient visited her pediatrician repeatedly, and began visiting neurologists, ear, nose and throat specialist (ENT''s), gastroenterologists (GI), and an obstetrics and gynecology (OBGYN) specialist, and made several visits to the Emergency Room. The patient received many blood tests, computerized tomogram (CAT) scans and a magnetic resonance imaging (MRI), but there was no relief of the patient''s symptoms. The reporter stated that the drugs prescribed to alleviate her symptoms only made her feel worse. The patient''s pediatrician and other doctors involved with the patient care began suggesting that her illness was psychosomatic and recommended to take her to a psychologist. In October 2010, the patient was first tested for Lyme disease and the results were negative (as were two later rounds of testing, exact dates unspecified). In April 2011, the patient could no longer go to school or participate in dance or cheerleading because the pain and fatigue were all consuming. The reporter stated that there was no relief of the patient''s symptoms and every doctor refused to consider Lyme disease or GARDASIL to be related to the patient''s illness. In approximately April 2011, the patient''s list of symptoms included the following: 24/7 headaches and migraines, 24/7 stabbing ear pain, hyperacusis, fatigue, abdominal pain, nausea, all over joint pain, constant sore throat, visual disturbances, light sensitivity, cognitive issues such as memory and severe comprehension problems, random numbness and tingling, weird "bug crawling" skin sensations, generalized weakness throughout her body (it was difficult for the patient to just sit in the shower to bathe), dizziness, fainting and heart palpitations. The patient slept long hours and stayed in her bedroom shielding herself from the noise of everyday living. In May 2011, tick-borne specialized lab tests were performed. The results showed that the patient was highly positive for Bartonella henselae, also highly positive for Mycoplasma pneumoniae and that her immune system was struggling. The patient''s Western Blot for Lyme disease was negative. The reporter mentioned that the results were taken to the local hospital and also to the patient''s neurologist, and even the patient had the classic Bartonella rash (looked like purple and red stretch marks) surrounding her breasts and hips which was confirmation of an active Bartonella infection, both doctors told her that these test results only showed that the patient was "exposed" to Bartonella but it did not mean she had an active infection. and that both came to the same conclusion that her Bartonella rash was actually just stretch marks. The reporter stated that neither doctor was concerned about the patient''s blazing Mycoplasma pneumoniae infection nor was the fact that the testing showed her immune system impaired. Instead, the hospital diagnostic center diagnosed the patient with the beginning stages of dysautonomia with no cure and with symptoms to be managed with medications. The hospital neurologist was not in agreement with the diagnostic center and diagnosed Chronic Migraine Disorder with Chronic Ear Pain Neuralgia. The neurologist recommended to continue with the same treatment of "20 pills a day" even though it did not improve the patient''s symptoms but increased her nausea and head pain. In June 2011, after nine months, the patient visited a Lyme Literate Medical Doctor (LLMD) who review her blood tests and other medical reports, and then clinically diagnosed her with Lyme disease, and agreed with the test results that reported active infections with Bartonella Henselae and Mycoplasma pneumoniae. The physician said the patient was very sick and acknowledged she was ill. Since that time, the patient was diagnosed with chronic Strep, human herpes virus 6 (HHV6), hypocoagulation, susceptibility to biotoxin illness (mold and environmental sensitivities) and had acquired autoimmune thyroid disease. The reporter stated that the patient was still chronically ill, she was unable to attend school in 7th and 8th grades, and she decided to repeat 8th grade again rather than begin high school still sick. The patient had a plan in school to reduce her daily schedule, she was still too sick to attend school with any regularity. Most days, a teacher went to her home to review the lessons she missed at school. Some days, the patient''s pain levels were too high so that she could not even tolerate home tutoring. Most days, the patient remained at her room sleeping and trying to cope with chronic pain. The reporter also stated that the LLMD and the patient''s Primary Care Physician reviewed the patient''s vaccine log and extensive medical records and both agreed that the patient''s immune system was injured by GARDASIL and that it was the catalyst to her cascading health problems and chronic illness. The patient''s LLMD was still treating her for a vaccine injury in addition to treating multiple tick-borne diseases, other infections/viruses and autoimmune thyroid disease. At the time of the report, the outcomes of the events Cat scratch disease, mycoplasma infection, immune system disorder, ear pain, pain, somnolence, neuralgia, autonomic nervous system imbalance, migraine, malaise, fatigue, and asthenia were assessed as not recovered/not resolved .The outcome for the other reported events was unknown. The reporter considered the all the reported events to be related to GARDASIL. Upon internal review, the events Cat scratch disease, mycoplasma infection, and Lyme disease were considered to be disabling because the patient was unable to attend school in 7th and 8th grades and was still too sick to attend school with any regularity. This case in linked to case MARRS# 1503USA015062 (same reporter link). Additional information is not expected because the reporter details were not provided.
新聞の見出し
「今危険性が問題となっているガーダシルを接種したエセックス、クラクトン在住のニコルさん
副反応のため、趣味のダンスを諦めなくてはならなかった
彼女のストーリーは、世界中で何千人もの少女たちが声をあげている副反応の症状と同じものである
しかし、WHOはそのような重篤な副反応があることを強く否定している
研究は、この議論の余地があるワクチンが効果的で安全であることを示している」
ガーダシル後、麻痺を発症
関節炎、胃腸の問題、ヘルニア、口腔内潰瘍、ポッツ、痛み、疲労、記憶障害、歩行障害などの症状、
ニコルさんは、ある朝テレビをみていて、副反応の症状の長いリストを聞き、ほとんどが自分の症状に当てはまり、自分の病気の原因がわかった
UKの被害者の会にはいり、自分と同じ立場の人と話ができたことで、自分のことを理解してもらえほっとしているとインタビューに答えている
甲状腺がんが増えているのはどうしてですか?

英国女性


アメリカ https://www.cancer.org/latest-news/4-essential-cancer-charts-for-2016.html
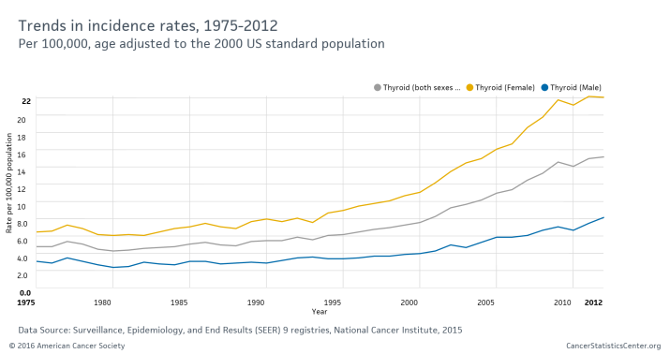

アメリカ

20~26歳女性
赤:ワクチン接種群 177人
青:ワクチン非接種群 701人
ワクチン接種群では、ワクチンに含まれる型以外のハイリスクHPV感染が増加している。
これって本当でしょうか?
不活化ワクチンを接種すると、特に母乳で育っていない乳児では免疫系がTh2優位となり、その後に生ワクチンを接種すると、生ワクチンに反応すべきTh1免疫系が弱いので、副反応が起きやすくなる
中津川亮さんがリツイート
A baby in Bomet county in Kenya died earlier this week shortly after receiving the measles vaccine at a government health center in the town of Mogogosiek. Four other babies—all under one year of age—were also given the measles vaccine at the health center and became swollen and critically ill. They were rushed to the intensive care and high dependency units at Tenwek Mission Hospital in Bomet.
英国のテレサ・メイ首相が、子宮頸がん検診の開始年齢を25歳から引き下げるよう国会で演説しました
子宮頸がんワクチン接種率が85%前後の英国で、接種世代の子宮頸がんが確実に増加していることを反映しています
英国でのサーバリックス導入は2008年。当時18歳だった被接種者は、今27歳になっています。
Cervical Cancer (C53), European Age-Standardised Incidence Rates, Females, by Age, UK, 1993-2014

2018-01-24
国際シンポジウム「世界のHPVワクチン被害は今」
HPVワクチン(「子宮頸がんワクチン」)によって深刻な被害が発生し、社会問題化しているのは日本だけではありません。本シンポジウムでは、集団訴訟が提起されているコロンビアを始め、スペイン、英国、アイルランドの被害者団体からゲストを迎え、被害実態、医学界や政府の対応、被害者の活動などを共有し、問題の本質や被害救済について議論します。是非ご参加ください。
■日時 2018年3月24日(土)13:30~17:30(13:00開場)
■会場 東京大学本郷キャンパス 武田先端知ビル 5F 武田ホール
■同時通訳付・事前申込不要・資料代500円
<プログラム>
■第1部 被害実態等-各国からの報告
コロンビアからの報告 Monica Leon Del Rio
"Rebuilding Hope Association HPV Vaccine Victims"代表、集団訴訟代理人弁護士
スペインからの報告 Alicia Capilla
"Association of Affected People due to the HPV vaccines in Spain"(AAVP)の代表
イギリスからの報告 Mandeep Badial
"UK Association of HPV Vaccine Injured Daughter"(AHVID)の科学部門担当
アイルランドからの報告 Anna Cannon
"Reactions and Effects of Gardasil Resulting in Extreme Trauma"(REGRET)広報担当
日本からの報告 水口真寿美
薬害オンブズパースン会議事務局長、HPVワクチン薬害訴訟全国弁護団代表
(※海外からの報告者は、いずれの方も被害者の母です)
■第2部 パネルディスカッション
パネリスト 第1部報告者他
コーディネーター 隈本邦彦他(薬害オンブズパースン会議メンバー)
主催 薬害オンブズパースン会議
問合せ先 薬害オンブズパースン会議事務局 TEL 03-3350-0607
e-mail yakugai@t3.rim.or.jp
URL http://www.yakugai.gr.jp
https://www.youtube.com/watch?v=Hgmjie_GcUE
フロリダ州の小児科医 医師歴30年
小児が必要とするワクチンだけを、適切なタイミングで接種している
ガーダシルは、発売当初、5人の少女に接種したが、5人ともごく軽症の副反応があったので
その後は、ガーダシルは接種していない (6分20秒くらいから)

麻疹のワクチンは3歳以降で接種
麻疹の罹患による重症化はビタミンAの欠乏によると証明されている
Autoimmune diseases, including multiple sclerosis and type 1 diabetes mellitus, affect about 5% of the worldwide population. In the last decade, reports have accumulated on various autoimmune disorders, such as idiopathic thrombocytopenia purpura, myopericarditis, primary ovarian failure, and systemic lupus erythematosus (SLE), following vaccination. In this review, we discuss the possible underlying mechanisms of autoimmune reactions following vaccinations and review cases of autoimmune diseases that have been correlated with vaccination. Molecular mimicry and bystander activation are reported as possible mechanisms by which vaccines can cause autoimmune reactions. The individuals who might be susceptible to develop these reactions could be especially not only those with previous post-vaccination phenomena and those with allergies but also in individuals who are prone to develop autoimmune diseases, such as those with a family history of autoimmunity or with known autoantibodies, and the genetic predisposed individuals.
Further research is encouraged into the direct associations between vaccines and autoimmune conditions, and the biological mechanisms behind them.
The HPV vaccines (such as Gardasil® and Cervarix®) were introduced to fight the cervical cancer; however, several cases of onset or exacerbations of autoimmune diseases following vaccination have been reported [128]. In 2013, Colafrancesco reviewed three women (two of them are sisters, thus bringing the relevance of genetics linkage) that developed primary ovarian failure within 2 years by HPV vaccine [129]. All the patients developed secondary amenorrhea, low estradiol, and high follicle-stimulating hormone (FSH) and luteinizing hormone (LH) following HPV vaccination, and elevated anti-antibodies levels (e.g., anti-thyroid antibodies and anti-ovarian antibodies). The authors suggested that the use of adjuvants in the HPV vaccine could be a risk factor for eliciting an autoimmune reaction to the vaccination: the DNA fragments detected in 16 different Gardasil® vaccines appeared to be bound to the aluminum used in the vaccine formulation.
In 2013, Gatto and coworkers investigated cases of SLE in women following HPV vaccination [130]. The onset of SLE occurred during the later doses of the HPV vaccination schedule and all the women had family histories of autoimmune disease. The major remitting patients with immunosuppression therapy had mild adverse effects to the vaccine immediately following the first dose of the HPV vaccine and then developed heavier SLE symptoms within two months after subsequent vaccine administration. In conclusion, the authors hypothesized a potential causal link between HPV vaccination and onset or relapse of SLE [130]. Thus, although for most patients, the benefits of immunization outweigh its risks, clinicians must be aware of the odds for an autoimmune disease onset or exacerbation following HPV vaccination. A meticulous pre-vaccination risk–benefits assessment, close follow-up during and after each boost of vaccination, as well as assessment of concomitant therapy with immune-modulating agents such as hydroxychloroquine (HCQ), seems reasonable for patients with an autoimmune disease. In 2013, Macartney and colleague reviewed the literature of HPV vaccine-related adverse effects and they reported almost mild reaction such as local injection site swelling with local or generalized pain [131]. Subjective reports are the primary source of vaccine adverse event but a systematic approach to track vaccination morbidity is required. In addition, the short follow-up institutionally fixed by the health authorities miss mild and severe long-term adverse reactions and large genetically different vaccinated group would better outline the problem. Thus, long-term surveillance of vaccines among interethnic populations groups would define more accurately their safety.
Recently, several reports have suggested grouping different autoimmune conditions that are triggered by external stimuli (e.g., exposure to vaccine) as a single syndrome called autoimmune syndrome induced by adjuvants (ASIA) [132]. This syndrome is characterized by the appearance of myalgia, myositis, muscle weakness, arthralgia, arthritis, chronic fatigue, sleep disturbances, cognitive impairment, and memory loss. This term was introduced by Yehuda Shoenfeld, who highlighted the pathogenic role of adjuvants in the induction of autoimmune syndromes: these compounds mimic evolutionarily conserved molecules (e.g., bacterial cell walls, LPS, unmethylated CpG-DNA) and bind to toll-like receptors (TLRs). They activate dendritic cells (DCs), lymphocytes, and macrophages, increasing subsequently the release of chemokines and cytokines from T-helper and mast cells [133–137]. The adjuvants added into vaccines can induce a non-specific activation of the immune system with a subsequent expansion of autoreactive (in our case, myelin specific) lymphocytes that may be further accelerated by defective regulatory cells/circuits, in genetically susceptible individuals. Indeed, the main individuals at ASIA syndrome risk are as follows: (1) patients with prior post-vaccination autoimmune phenomena, (2) patients with a medical history of autoimmunity, (3) patients with a history of allergic reactions, and individuals who are prone to develop autoimmunity (having a family history of autoimmune diseases, presence of autoantibodies, carrying certain genetic profiles, etc.) [138].
In the 2016, we retrospectively described a case series including 18 girls (aged 12–24 years) for the evaluation of “neuropathy with autonomic dysfunction” immediately after HPV vaccine [Gardasil® (9 girls) and Cervarix® (9 girls)] [139]. All girls complained of long-lasting and invalidating somatoform symptoms of the recently described ASIA syndrome (including asthenia, headache, cognitive dysfunctions, myalgia, sinus tachycardia, and skin rashes) that have developed 1–5 days (n = 11), 5–15 days (n = 5), and 15–20 days (n = 2) after the last dose vaccination. The HPV vaccine formula contains aluminum (225 e 500 μg/each dose in Gardasil® and Cervarix®, respectively) but also high polysorbate 80 (50 mcg) concentration that might also induce a greater meningeal permeability leading to a facilitated entrance of many substances to the CNS. Based upon these observations, it might be speculated that HPV vaccine could induce some abnormal activation of immune competent cells in the CNS, such as the glia.
Among the risk factors for ASIA syndrome, the metal hypersensitivity in girls, mainly genetically predisposed individuals, exposed to immunization, has been suspected. We tested five adjuvant metals (aluminum, mercury, nickel, methyl-mercury, thimerosal) in our case series through in vitro blood test and lymphocyte transformation test (MELISA®). However, the study was frustrating, being the seven girls negative to each of the five metals tested, showing a metal-hypersensitivity only in nine patients: toxicity to aluminum (two girls), reactivity to nickel (seven girls), followed by mercury (four girls) [140].
Case control and epidemiological studies and a detailed genetic analysis of affected girls and their family might better define the link between vaccination and CNS damage.
Transverse myelitis (TM) is the paradigm of inflammatory myelopathy, in which an immune-mediated process causes neural injury to the spinal cord, resulting in varying degrees of weakness, sensory alterations, and autonomic dysfunction. TM may exist as part of a multifocal central nervous system disease (e.g., multiple sclerosis), multisystemic disease (e.g., SLE), or as an isolated idiopathic entity [141]. A recent review (2014) summarizes nine published cases of CNS demyelination, including not only myelitis but also optic neuritis and encephalitis, following HPV vaccination [142]. There are previous reports of CNS inflammatory syndromes following HPV vaccination describing a 10-day to 5-month time lapse from vaccination to symptom onset, with a minimum of a 21-day interval in cases developing myelitis. For instance, a 14-year-old immunocompetent girl noticed her left hand felt weak and numb, after 3 days of a first dose of HPV vaccine (Gardasil®) and these symptoms spread to the rest of her arm and ipsilateral thoracic region, abdomen, and leg. On a medical examination, the girl had bilateral diminished sensation to light touch, pain, and vibration below C5 level; muscle weakness of her left arm and leg; and hyperreflexia bilaterally with nonsustained clonus and a left extensor plantar response. Neurologic examination was otherwise normal. The relationship between HPV vaccination and subsequent CNS inflammation remains unclear. In the genesis of CNS inflammatory disorders post-HPV vaccination, both molecular mimicry between vaccine antigen and myelin proteins and toxic materials in vaccine components can represent potential causative factors [143]. Regarding etiology, the patient was immunocompetent and there were no systemic signs of infection. The absence of cells in CSF analysis and the presence of oligoclonal bands with an increased nonspecific immunoglobulin M ratio suggested a background of a CNS inflammatory condition; however, the mechanism of disease, whether viral-induced or immune-mediated, remains to be determined.
グーグル翻訳で訳してみました
WHOのVigiAccessに報告されたHPVワクチン後の免疫系障害 カッコ内は報告数
免疫系障害(1741)
過敏症(651)
アナフィラキシー反応(323)
自己免疫障害(227)
免疫系障害(115)
食物アレルギー(82)
アナフィラキシーショック(75)
免疫応答の低下(41)
薬物過敏症(41)
季節性アレルギー(34)
免疫反応(32)
血清病(32)
ワクチンへのアレルギー(28)
アナフィラキシー様反応(26)
免疫不全(16)
複数のアレルギー(14)
ミルクアレルギー(11)
動物アレルギー(9)
金属アレルギー(9)
III型免疫複合体媒介反応(9)
免疫抑制(8)
真菌性アレルギー(8)
ダニアレルギー(7)
香水の感度(7)
防腐剤に対する反応(7)
タイプI過敏症(7)
複数の化学的感受性(6)
植物アレルギー(4)
腎臓移植拒絶反応(4)
口腔アレルギー症候群(4)
賦形剤(4)に対する反応
アトピー(3)
多発性血管炎による好酸球性肉芽腫症(3)
低貧血症(3)
ゴム感受性(3)
サルコイドーシス(3)
節足動物の咬傷に対するアレルギー(2)
カフェインアレルギー(2)
低γグロブリン血症(2)
免疫不全共通変数(2)
食品添加物に対する反応(2)
選択的IgA免疫不全(2)
血清病様反応(2)
タイプIV過敏反応(2)
ABOの非互換性(1)
アレルギー性浮腫(1)
化学物質へのアレルギー(1)
アナフィラキシーショックショック(1)
抗好中球細胞質抗体陽性脈管炎(1)
自己炎症性疾患(1)
免疫再構成炎症性症候群(1)
重複症候群(1)
煙感受性(1)
移植拒絶反応(1)
II型過敏症(1)
グーグル翻訳で訳してみました
WHOのVigiAccessに報告されたHPVワクチン後の腫瘍など カッコ内は報告数
新生物は良性、悪性および不特定(嚢胞およびポリープを含む)(1005)
肛門性器疣贅(261)
皮膚乳頭腫(151)
子宮頸がん(87)
子宮頸部癌ステージ0(65)
ホジキン病(38)
悪性新生物(30)
in situ癌(25)
急性骨髄性白血病(15)
白血病(15)
急性リンパ球性白血病(13)
卵巣癌(12)
子宮膣疣贅(12)
脳腫瘍(11)
メラノサイトネイバス(11)
非ホジキンリンパ腫(11)
甲状腺癌(11)
リンパ腫(10)
新生物(10)
扁平上皮癌(9)
血管腫(8)
子宮頸部腺癌(7)
乳癌(7)
喉頭乳頭腫(7)
乳房線維腫(6)
組織壊死性リンパ節炎(6)
パピローマ(6)
呼吸器パピローマ(6)
子宮平滑筋腫(6)
子宮頸部癌II期(5)
転移(5)
骨肉腫(5)
下垂体腫瘍良性(5)
うずまき癌(5)
腺癌(4)
子宮頸癌ステージI(4)
慢性骨髄性白血病(4)
ホジキン病II期(4)
骨髄異形成症候群(4)
口腔乳頭腫(4)
甲状腺乳頭腫(4)
アクロコドン(3)
骨がん(3)
焦点性結節性過形成(3)
ホジキン病結節性硬化症(3)
脂肪腫(3)
新生物の皮膚(3)
卵巣胚細胞奇形腫(3)
下垂体腫瘍(3)
原発性縦隔大B細胞リンパ腫(3)
横紋筋肉腫(3)
肉腫(3)
子宮癌(3)
急性白血病(2)
急性前骨髄球性白血病(2)
肛門癌ステージ0(2)
良性の水胞子(2)
骨肉腫(2)
脳新生物良性(2)
悪性脳腫瘍(2)
脳幹神経膠腫(2)
女性の乳癌(2)
子宮頸がんステージIV(2)
皮膚線維肉腫プロトプランズ(2)
デスモイド腫瘍(2)
子宮内膜がん(2)
アイネイバー(2)
骨の血管腫(2)
肝がん(2)
大粒リンパ球増加症(2)
喉頭がん(2)
肺新生物(2)
リンパ管腫(2)
縦隔腫瘍(2)
肝転移(2)
骨髄性白血病(2)
転移性膵臓内分泌腫瘍(2)
パイオニア肉芽腫(2)
子宮頚部の扁平上皮癌(2)
甲状腺新生物(2)
卵巣癌ステージ0(2)
腹部新生物(1)
腺腫良性(1)
未分化大細胞リンパ腫T細胞およびヌル細胞型(1)
B細胞リンパ腫(1)
B細胞型急性白血病(1)
良性骨新生物(1)
良性乳房新生物(1)
良性肺新生物(1)
良性リンパ節新生物(1)
良性新生物(1)
良性の脊髄新生物(1)
良性卵巣腫瘍(1)
良性子宮腫瘍(1)
尋常性乳頭腫(1)
骨新生物(1)
現場乳癌(1)
寛解の癌(1)
中枢神経系リンパ腫(1)
中枢神経系新生物(1)
子宮頸部癌III期(1)
胆嚢癌(1)
結腸癌(1)
女性の生殖腫瘍(1)
フィブロマ(1)
消化管新生物(1)
生殖器新生物悪性女性(1)
グリオーマ(1)
肝臓血管腫(1)
皮膚の血管腫(1)
肝腺腫(1)
肝芽腫(1)
ホジキン病結節性硬化症II期(1)
ホジキン病再発(1)
ホジキン病IV期(1)
インスリノーマ(1)
寛解期の白血病(1)
口腔および/または口腔癌(1)
肺悪性新生物(1)
肺扁平上皮癌IV期(1)
リンパ系新生物(1)
リンパ球性白血病(1)
リンパ組織球増多症(1)
リンパ増殖性障害(1)
悪性縦隔新生物(1)
悪性黒色腫ステージII(1)
悪性呼吸器腫瘍(1)
髄芽腫(1)
心臓転移(1)
腎臓への転移(1)
リンパ節転移(1)
転移性新生物(1)
菌状息肉腫(1)
ニューロマ(1)
眼球新生物(1)
卵巣癌ステージIII(1)
卵巣生殖細胞テラトーマ良性(1)
卵巣胚細胞腫瘍(1)
卵巣顆粒膜細胞腫瘍(1)
膵臓癌(1)
乳頭結膜(1)
悪性腫瘍症候群(1)
真珠の陰茎の丘疹(1)
陰茎のいぼ(1)
末梢T細胞リンパ腫不明(1)
再発癌(1)
肉腫転移性(1)
皮膚がん(1)
軟部肉腫(1)
スピンドル細胞肉腫(1)
脾腫新生物悪性腫瘍(1)
ドイツから
ワクチン接種前に性交経験なしの14歳の少女がガーダシル接種後、2年後に子宮頸がん発症。
| VAERS ID: | 528233 (history) |
| Form: | Version 1.0 |
| Age: | 14.0 |
| Gender: | Female |
| Location: | Foreign |
| Vaccinated: | 0000-00-00 |
| Onset: | 0000-00-00 |
| Submitted: | 2014-04-11 |
| Entered: | 2014-04-11 |
| Vaccination / Manufacturer | Lot / Dose | Site / Route |
| HPV4: HPV (GARDASIL) / MERCK & CO. INC. | - / 3 | UN / UN |
Administered by: Other Purchased by: Other
Symptoms: Cervix carcinoma, Vaccination failure
SMQs:, Lack of efficacy/effect (narrow), Uterine and fallopian tube malignant tumours (narrow), Non-haematological malignant tumours (narrow)
Life Threatening? No
Birth Defect? No
Died? No
Permanent Disability? No
Recovered? No
Office Visit? No
ER Visit? No
ER or Doctor Visit? No
Hospitalized? No
Previous Vaccinations:
Other Medications: No other medications
Current Illness: Immunisation
Preexisting Conditions: GARDASIL, Immunisation; GARDASIL, Immunisation
Allergies:
Diagnostic Lab Data:
CDC Split Type: WAES1404DEU006071
Write-up: Case of vaccination failure was received from a physician (also reported other health professional) via a sales representative on 06-Apr-2014 from Sanofi Pasteur MSD (reference # DE-1577272925-E2014-03061) on 09-APR-2014. Case is medically confirmed and poorly documented. An approximately 14-year-old female patient received a complete vaccination series with three doses of GARDASIL (lot no., route and site of administration not reported) on unspecified dates. Two years later, at the age of 16, she developed cervical carcinoma. Duration and outcome were not reported. Vaccination breakthrough was concluded by the reporter. The patient had no sexual intercourse before vaccination.
アメリカのカリフォルニア州で、インフルエンザが猛威を奮っていると、ジェームズ・マドックCEO率いる21世紀フォックス系メディアが報道しているそうで、カリフォルニアの救急では、患者が殺到し、対処しきれずに家に送り返されたり、また、他の州から応援の看護師が大量に送り込まれているということです。
このニュースを聞いたカリフォルニア在住のジャーナリストが実際にニュースで言及された複数の病院に行ったところ、どこでも救急病棟は閑散としていたと報告しています。
(情報源 ビデオhttps://www.youtube.com/watch?v=F_pO8ai1WDI)
フェイクニュースだったのか?!
日本でも、インフルエンザ様ワクチンで、9月の新学期早々学級封鎖があったようですが、こちらのフェイク度はどうでしょうか?
http://www.kyoiku.metro.tokyo.jp/press/2017/pr170905b.html

幸い、他の学級・学校には波及しなかったようです。
その後の東京都のインフルエンザ様疾患の報告は以下のサイトの下記のグラフから
http://idsc.tokyo-eiken.go.jp/diseases/flu/flu/
学級閉鎖が12月の後半に集中しているのは、佐賀県でもみられました。
http://kansen.pref.saga.jp/influenza/heisa4/171219.pdf
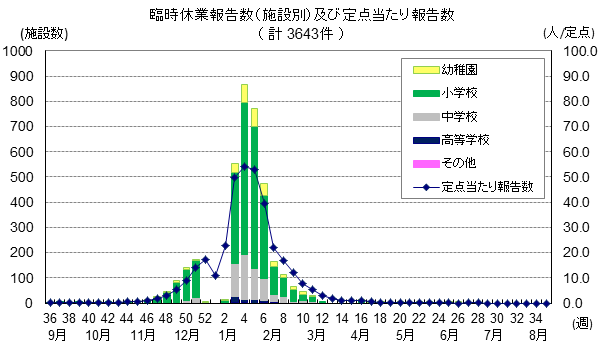
定点当たり報告数が増加しているので、今週、来週あたりは、東京都でも学級閉鎖が増加するのでしょうか。