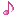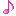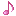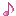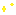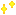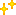Reactions (BiO-BrO) εo (V vs. SHE) Ref.
Bi2O3 + 6H+ + 6e ↔ 2Bio + 3H2O -0.46(1), 0.376(2)
Bi2O4 + 4H+ + 2e ↔ 2BiO+ + 2H2O 1.593 1
BiO+ + 2H+ + 3e ↔ Bio + H2O 0.320 1
[BiOH]2+ + 3e ↔ Bio + OH- 0.072 2
BiOCl + 2H+ + 3e ↔ Bio + Cl- + H2O 0.1583(1), 0.1697(2)
Bk4+ + e ↔ Bk3+ 1.67 1, 2
Bk3+ + e ↔ Bk2+ -2.8 1
Bk3+ + 3e ↔ Bko -2.01 2
Bk2+ + 2e ↔ Bko -1.67 1
Br2(aq) + 2e ↔ 2Br- 1.0873(1), 1.0874(2)
Br2(l) + 2e ↔ 2Br- 1.066(1), 1.0652(2)
Br3- + 2e ↔ 3Br- 1.0503 2
HBrO + H+ + 2e ↔ Br- + H2O 1.331 1
HBrO + H+ + e ↔ 1/2Br2(aq) + H2O 1.574 1
HBrO(aq) + H+ + e ↔ 1/2Br2(l) + H2O 1.596(1), 1.604(2)
BrO- + H2O + 2e ↔ Br- + 2OH- 0.761 1
BrO- + H2O + e ↔ 1/2 Br2(l) + 4OH- 0.455 1, 2
BrO3- + 5H+ + 4e ↔ HBrO(aq) + 2H2O 1.447 2
BrO3- + 6H+ + 5e ↔ 1/2Br2 + 3H2O 1.482 1
BrO3- + 6H+ + 6e ↔ Br- + 3H2O 1.423 1